Disclaimer: This document is intended as a resource for health care providers caring for COVID-19 patients, and is based on available scientific evidence and scientific guideline writing committee/ expert documents as well as our own opinions. These recommendations do not replace clinical judgment or the need for individualized patient care plans. While we attempt to keep this document up-to-date, the literature on COVID-19 is rapidly evolving, and we suggest that practitioners search for the most up-to-date literature on any specific topic. Finally, these guidelines were developed based on health care practices at the University of California San Francisco in San Francisco, California and may not reflect these factors in other health care systems and in other geographic locations.
University of California, San Francisco Cardiology
Covid-19 Clinical Protocols and Guidelines
Cardiac ICU Considerations for SARS-CoV2
Cardiac Inpatient Considerations in COVID+ Patients
Transthoracic and Transesophageal Echocardiograms
Treatment With Renin-Angiotensin System (RAS) Blockers
Adult Congenital Heart Disease
UCSF Infectious Disease COVID-19 Guidelines (link)
Cardiac ICU Considerations For SARS-CoV2
Purpose:
There is a high prevalence of underlying cardiovascular disease comorbidities in patients hospitalized with COVID-19 infection and these patients are at increased risk of mortality with COVID-19 infection1,2. Further evidence of acute cardiac dysfunction on presentation with COVID-19 disease is also linked with disease mortality3,4. Case reports have described acute cardiac involvement with COVID-19 infection, although the true incidence of this complication is unknown5. But, with increasing prevalence of COVID-19 disease in the general population, we expect to see patients presenting with simultaneous COVID-19 infection and acute exacerbations of underlying cardiac disease. These guidelines are intended to supplement existing UCSF critical care guidance. A brief discussion of management of infectious concerns in COVID patients as well as techniques for management of ARDS and hypoxic respiratory failure is included. Specific attention is paid to the unique concerns of patients with critical cardiac illness.
General Principles:
1. Given rapidly changing knowledge about this disease, the guidance in this document will require revision and update. We will attempt to keep the guidance as up to date as possible. The version number and date are noted in the header.
2. Patients with cardiovascular disease often require bedside procedures which require increased monitoring and exposure.
3. In managing COVID-19 patients it is important to minimize exposure and conserve PPE.
4. Management of Respiratory Failure with coexisting cardiovascular disease requires particular care.
Procedures:
General Principles for Bedside Procedures:
When possible, patients requiring invasive procedures for either hemodynamic monitoring, support, or pacing should be performed at the bedside to minimize patient transport and the risks of exposure during transport. Bedside procedures require the following considerations:
Precautions: Thus far, there are no reports of bloodborne transmission of the SARs-CoV2 virus. Viral RNA can be isolated in patient blood and so transmission from direct exposure of infected blood is possible. During bedside procedures universal precautions including gloves, mask, and eye protection should be employed. Individual procedures will be noted as to whether or not the procedure is considered aerosol generating. However, even non-aerosol generating procedures may require the patient to lay flat for an extended period of time and can be associated with respiratory decompensation. For patients with tenuous respiratory status, consideration of pre procedure intubation should be made as well as preparation for urgent intubation during the procedure.
Personnel: Bedside procedures should be performed by skilled operators who are comfortable with intravascular access, obtaining hemostasis, and bailout maneuvers for complications including vascular damage. The bedside nurse should present to provide non-sterile assistance to the operator. An additional support staff should be present outside the room to obtain any additional supplies that are needed or obtain additional help should it be needed during the procedure. This staff member should wear PPE as well.
Indication: Given the risk of inadvertent contamination and exposure with prolonged contact, it is appropriate to use judicious clinical judgment prior to attempting bedside procedures. Individual clinical judgment will be essential in evaluating necessity of clinical decisions. Unindicated procedures should not be performed regardless of COVID-19 status.
1. Invasive Hemodynamic Monitoring:
In patients with undifferentiated shock, invasive arterial and central venous monitoring can be helpful in diagnosing the shock state and establishing ventricular filling pressures. Invasive monitoring of cardiac output is particularly helpful to guide the use of inotropic medications. In general, the Department of Cardiology advocates against the empiric use of inotropic medications without objective measurements of cardiac output. These are dynamic measurements that should be evaluated to titrate therapies or evaluate change in clinical stability.
- Central lines (TLC): Central line catheter placement has a role for frequent blood draws, infusion of vasoactive substances, hemodialysis, and invasive hemodynamic monitoring. In patients with undifferentiated shock, measurement of central venous gases can be useful in diagnosis of the shock state. Outside of clinical cardiogenic shock, use of central venous gas to guide resuscitation is unsupported by evidence.
- Pulmonary Artery Catheter (PAC): In patients with evidence of reduced cardiac output and shock, PAC placement may be helpful to guide use of inotropic agents and management of volume status. Rarely there is a role for PAC placement for the evaluation of unexplained hypoxia or to clarify left ventricular filling pressures in the setting of unexplained pulmonary infiltrates.
Personnel:
a. Operator: When performed at bedside, PAC placement should be performed by an experienced operator who is comfortable with vascular access and management of vascular complications. The operator should also be comfortable with the interpretation of hemodynamic waveforms.
b. Bedside nursing: The bedside nurse will be required to provide non-sterile assistance for vascular access and adjustment of the hemodynamic monitor.
c. Bedside fluoroscopy may be helpful if PAC placement using pressure monitoring is not successful. The radiation technician should wear appropriate PPE for the procedure.
2. Temporary Mechanical Circulatory Support:
IABP:
a. Procedure: IABP may be placed in either the operating room (OR), the cardiac catheterization laboratory, or at bedside. If placed at the bedside, consider portable fluoroscopy. IABP placement is not an aerosol generating procedure.
b. Monitoring: Vascular pulses should be assessed in the bilateral lower extremities and the left upper extremity per unit protocol[KB2] . Access sites should also be assessed every 8 hours. The IABP console should be turned towards the door with the waveform easily visible from outside the room.
Impella:
c. Procedure: Impella (CP, 5.0, or RP) should be placed in either the hybrid OR or the catheterization lab using fluoroscopy. Impella placement is not an aerosol generating procedure.
d. Monitoring: Vascular pulses should be assessed in the bilateral lower extremities extremities per unit protocol. Access sites should also be assessed every 8 hours. The Impella console should be turned towards the door with the waveform easily visible from outside the room. Bloodwork should be monitored as routine.
ECMO:
e. Please see guidance document for ECMO here.
3. Temporary Pacing Wires:
Patients with critical cardiac illness will occasionally require temporary transvenous pacing. When possible the procedure should be done at the bedside to minimize inadvertent exposure.
a. Operator: When performed at bedside, transvenous pacemaker placement should be performed by an experienced operator who is comfortable with vascular access and management of vascular complications. The operator should also be comfortable with catheterization of the right ventricle and settings of a temporary VVI pacemaker.
b. Bedside nursing: The bedside nurse will be required to provide non-sterile assistance for vascular access and adjustment of the pacemaker settings during placement.
c. Bedside fluoroscopy may be helpful if temporary pacing wire placement is not successful. The radiation technician should wear appropriate PPE for the procedure.
4. Transesophageal Echocardiography:
Please see current echocardiography guidelines here
5. Endotracheal Intubation:
a. Endotracheal intubation is considered an aerosol generating procedure and requires airborne precautions.
b. Given that the risks of exposure and contamination are particularly high during intubation, personnel involved should be limited. This also ensures adequate PPE for providers performing endotracheal intubation
c. Please see guidance specific guidance for intubation here
6. CRRT:
Both patients with critical cardiac disease and patients with respiratory failure frequently benefit from a net negative fluid balance. Among patients with critical cardiac disease, cardiorenal dysfunction is common and renal replacement therapy is frequently used in the ICU setting. In these situations, ultrafiltration can be used to maintain a net negative fluid balance. CRRT should be utilized as clinically indicated per usual protocol.
Respiratory Support:
7. Non-invasive Respiratory Support:
General considerations: Patients with COVID-19 illness and cardiovascular disease have high likelihood of progression to respiratory failure. Patients managed with non-invasive respiratory support (HFNO or NIPPV) should be carefully monitored in a setting that has adequate PPE and staffing for endotracheal intubation if necessary.
a. Nasal Cannula:
i. Nasal cannula oxygen support is recommended for all patients as with usual care. This is not considered an aerosol generating procedure and does not require airborne precautions.
b. High Flow Nasal Oxygen (HFNO):
i. HFNO can be considered for selected patients with hypoxemia. There is limited data regarding the safety of HFNO and it should be considered an aerosol generating procedure and requires airborne precautions.
c. Non-invasive Positive Pressure Ventilation (CPAP and BiPAP):
i. NIPPV is considered an aerosol generating procedure and requires airborne precautions. NIPPV should only be used in select situations with careful monitoring.
d. Nebulizer Therapy:
i. Nebulizer therapy is considered an aerosol generating procedure. Meter-dose inhalers should be used instead. Nebulizers should be avoided unless there is strong evidence for benefit.
8. Mechanical Ventilation in ARDS:
Basic Principles: The most common complications of COVID-19 infection regardless of comorbid cardiovascular disease is development of viral pneumonia and subsequent acute respiratory distress syndrome (ARDS)1. ARDS is a process of inflammatory lung injury resulting in damage to the capillary endothelium and alveolar epithelium resulting capillary leak, non-cardiogenic pulmonary edema, and ultimately pulmonary fibrosis6. Practically, ARDS is defined by the Berlin Definition: 1) acute onset within 7 days, 2) bilateral opacities on radiography, 3) non hydrostatic (cardiogenic) etiology for pulmonary edema, 4) with severity determined by the degree of hypoxemia as determined by the PaO2/FiO2 ratio7. Therapies rely on maintaining adequate oxygenation and ventilation to avoid tissue hypoxemia and end organ damage while avoiding ventilator lung injury.
Lung Protective Ventilation: Previous studies have shown that a strategy of low tidal volume ventilation (LTVV) and higher positive end expiratory pressure (PEEP) improves survival with ARDs8–10.
a. Tidal volumes – 4-8 cc/kg (Predicted body weight )
b. PEEP and FiO2 are typically managed using the “ARDSNet Protocol” which attempts to minimize oxygen toxicity by balancing the fraction of inspired oxygen delivered with PEEP to maintain PaO2 between 55 and 80 or SpO2 between 88 and 95%.
c. Permissive hypercapnia: In an effort to maintain low tidal volumes, a respiratory acidosis is typically tolerated with an acceptable pH 7.25-7.5.
Cardiac Considerations: The application of positive pressure ventilation has effects not only on the respiratory system and oxygen extraction but also on the cardiovascular system and oxygen delivery. These effects are amplified in the case of patients with cardiovascular dysfunction with distinct effects on left and right ventricular function. The effects of positive pressure ventilation further depend hemodynamic state11.
d. Left ventricle (LV): In general, positive pressure ventilation and PEEP decrease left ventricular afterload by decreasing the transmural pressure gradient across the LV cavity wall. Simultaneously, positive pressure ventilation and PEEP decrease LV filling pressures by decreasing RV output and pulmonary venous return. In patients with reduced LV contractility, reductions in afterload typically increase stroke volume. The effect of preload reduction on LV function depends on volume status. In hypervolemic patients, reduction in left atrial pressure is beneficial. In hypovolemic patients, reduction in left atrial pressure can reduce cardiac output.
e. Right ventricle (RV): In general, positive pressure ventilation decreases systemic venous return and right ventricular filling pressures. The effects of positive pressure ventilation and PEEP on right ventricular afterload are more variable. Most often, increased intrathoracic pressure increases pulmonary vascular resistance and right ventricular afterload. The exception is in patients with significant atelectasis who are breathing at low lung volumes and in patients with hypoxia. Both low lung volumes and hypoxia can cause pulmonary vasoconstriction. To the extent that PEEP corrects either of these two conditions, positive pressure ventilation may decrease pulmonary vascular resistance and right ventricular afterload.
9. Prone Positioning:
In patients with severe ARDS and persistent hypoxia, prone positioning improves oxygenation by improving the ventilation/perfusion mismatch seen in ARDS. A single randomized trial has shown a mortality benefit to early prone positioning in selected patients with severe ARDS12. This mortality benefit has been supported in subsequent meta-analyses13–15.
a. Indications: Patients with refractory hypoxemia despite a strategy of lung protective ventilation. There should be a multidisciplinary discussion with nursing, respiratory therapy, critical care medicine, and cardiac intensive care teams prior to initiating prone positioning ventilation.
b. Contraindications: Spinal instability, traumatic fracture, acute hemorrhage, ongoing shock, elevated ICP, recent surgery. In most circumstances, patients requiring mechanical circulatory support are not candidates for prone ventilation. As such cardiogenic shock should be considered a relative but not absolute contraindication to prone positioning.
c. Considerations: Prone positioning is a labor-intensive maneuver that has risks of peri-maneuver hypotension and hypoxia. A member of the primary physician team should be present when moving the patient from prone to supine or vice versa to assist both physically and with potential hemodynamic or respiratory instability. The risk of exposure and inadvertent contamination should be weighed against the benefits of prone positioning. Once the patient is in the prone position, additional procedures are often precluded. As such, teams should consider the need for subsequent invasive procedures when making the decision to pursue prone positioning.
10. Rescue Therapies:
a. Paralysis: Neuromuscular blockade has been shown to be beneficial in patients with severe ARDS by reducing patient-ventilator dyssynchrony and improving oxygenation. Neuromuscular blockade requires adequate deep sedation prior to initiation and frequent monitoring of neuromuscular response by nursing.
b. Inhaled nitric oxide or epoprostenol: Some studies have shown that inhaled pulmonary vasodilators may improve pulmonary arterial pressures and oxygenation in patients with ARDS. However, these agents have not been shown to improve either mortality or morbidity in ARDS. It is important to recognize that in patients with elevated left atrial pressures, pulmonary vasodilators may result in pulmonary edema and worsening hypoxia. In these cases, use of a PAC to monitor left ventricular filling pressures with therapy may be helpful.
c. ECMO: See ECMO guidelines here.
Further Resources:
1. Surviving Sepsis Campaign: Guidelines on the Management of Critically Ill Adults with Coronavirus-19 Disease (COVID-19)
2. ELSO Guidance Document: ECMO for COVID-19 Patients with Severe Cardiopulmonary Failure
Discussed with and reviewed by representatives from:
- Cardiac Critical Care
- Advanced Heart Failure
References:
1. Guan W-J, Ni Z-Y, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. February 2020. doi:10.1056/NEJMoa2002032
2. Li B, Yang J, Zhao F, et al. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. March 2020:10.1007/s00392-020-01626-01629. doi:10.1007/s00392-020-01626-9
3. Guo T, Fan Y, Chen M, et al. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. March 2020. doi:10.1001/jamacardio.2020.1017
4. Shi S, Qin M, Shen B, et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. March 2020. doi:10.1001/jamacardio.2020.0950
5. Hu H, Ma F, Wei X, Fang Y. Coronavirus fulminant myocarditis saved with glucocorticoid and human immunoglobulin. Eur Heart J. March 2020. doi:10.1093/eurheartj/ehaa190
6. Piantadosi CA, Schwartz DA. The acute respiratory distress syndrome. Ann Intern Med. 2004;141(6):460-470. doi:10.7326/0003-4819-141-6-200409210-00012
7. Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: The Berlin definition. JAMA - J Am Med Assoc. 2012;307(23):2526-2533. doi:10.1001/jama.2012.5669
8. Brower RG, Matthay MA, Morris A, Schoenfeld D, Thompson BT, Wheeler A. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342(18):1301-1308. doi:10.1056/NEJM200005043421801
9. Putensen C, Theuerkauf N, Zinserling J, Wrigge H, Pelosi P. Meta-analysis: Ventilation strategies and outcomes of the acute respiratory distress syndrome and acute lung injury. Ann Intern Med. 2009;151(8):566-576. doi:10.7326/0003-4819-151-8-200910200-00011
10. Petrucci N, De Feo C. Lung protective ventilation strategy for the acute respiratory distress syndrome. Cochrane Database Syst Rev. 2013;2013(2):CD003844. doi:10.1002/14651858.CD003844.pub4
11. Gomez H, Pinsky MR. Effects of mechanical ventilation on heart-lung interactions. Princ Pract Mech Vent. 2012:1-31.
12. Guérin C, Reignier J, Richard JC, et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368(23):2159-2168. doi:10.1056/NEJMoa1214103
13. Sud S, Friedrich JO, Taccone P, et al. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: Systematic review and meta-analysis. Intensive Care Med. 2010;36(4):585-599. doi:10.1007/s00134-009-1748-1
14. Hu SL, He HL, Pan C, et al. The effect of prone positioning on mortality in patients with acute respiratory distress syndrome: A meta-analysis of randomized controlled trials. Crit Care. 2014;18(3). doi:10.1186/cc13896
15. Bloomfield R, Noble DW, Sudlow A. Prone position for acute respiratory failure in adults. Cochrane Database Syst Rev. 2015;2015(11):CD008095. doi:10.1002/14651858.CD008095.pub2
Cardiac Monitoring in COVID+ Inpatients:
Background:
There is increasing evidence that both preexisting cardiovascular disease and de novo cardiac injury are involved in the pathophysiology of COVID-19 infection1–3. Cardiovascular compromise is believed to play an important role in predicting the course and ultimate outcomes of COVID-19 infection4,5. Large cohort studies have demonstrated escalating severity of disease in patients with one or both of underlying cardiovascular disease and with acute cardiac injury. Further, case reports have described cardiac involvement in the presentation of the disease ranging from myocarditis, heart failure, and cardiogenic shock6,7 to unpublished reports of acute coronary syndrome and acute myocardial infarction. Additionally, with increasing prevalence of COVID-19 disease the general population, we expect to see patients presenting with simultaneous COVID-19 infection and acute exacerbations of underlying cardiac dysfunction.
These recommendations provide guidance in the cardiac evaluation of patients with COVID-19 disease and how cardiac assessment can be used for risk stratification of patients presenting with COVID-19 disease. Importantly, despite the frequency of acute myocardial injury in patients presenting with COVID-19 disease, the reported incidence of plaque rupture and acute coronary syndrome pathophysiology is unknown. The majority of acute cardiac injury in these patients is not felt to represent plaque rupture physiology however. Rather elevation in cardiac biomarkers is felt to hold prognostic value regarding the severity of infection with the underlying pathophysiology under ongoing investigation8. Patient evaluation for acute coronary syndrome remains unchanged relying primarily on a careful assessment of anginal symptoms in combination with biomarkers, electrocardiogram, and the overall clinical picture. Recommendations around the care of patients presenting with acute myocardial infarction including ST elevation myocardial infarction can be found here. These recommendations apply to the evaluation and risk stratification of patients presenting with COVID-19 disease.
Purpose:
The recommendations create a general framework for the cardiac evaluation and risk stratification of patients presenting with COVID-19 disease. Recommendations are stratified by patient status (Floor, TCU, and ICU), (what about ED?). Separate documentation regarding specific recommendations for CCU patients is available here. Lastly recommendations for specific patients with underlying cardiac disease are available at the end.
Inpatient Status | Initial Evaluation | Repeat Evaluation |
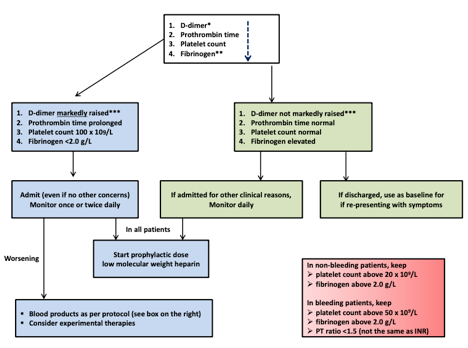
Floor/TCU Status | • History and physical with careful attention to cardiac risk factors (HTN, DM, CAD, CVA, or PAD) • Baseline bloodwork • Troponin I • BNP • D-Dimer, Fibrinogen, Platelet Count, Prothrombin time as per ISTH Interim Guideline Algorithm (see below) • 12 lead ECG • Echocardiography. See below for further guidance. | • Telemetry • Repeat troponin and BNP if clinically indicated by new symptoms of ischemia or ventricular arrhythmia • Repeat D-Dimer, Fibrinogen, Platelet Count, Prothrombin time as per ISTH Interim Guideline Algorithm |
ICU Status | • History and physical with careful attention to cardiac risk factors (HTN, DM, CAD, CVA, or PAD) • Baseline bloodwork • Troponin I • BNP • CK as per protocol with fractionation if doubling of troponin • D-Dimer, Fibrinogen, Platelet Count, Prothrombin time as per ISTH Interim Guideline Algorithm • ScVO2 for patients in undifferentiated shock • 12 lead ECG • Echocardiography. See below for further guidance. | • Telemetry • Q3 day repeat troponin and BNP if stable, sooner if change in clinical status • Repeat ScVO2 with change in clinical status • Repeat D-Dimer, Fibrinogen, Platelet Count, Prothrombin time as per ISTH Interim Guideline Algorithm |
Triggers for cardiology consultation:
Prior to cardiology consult, please obtain ECG, BNP, and troponin. If the consult is for shock please obtain ScVO2 and CVP as well. Cardiology will be able to assist in interpretation of the data. If the data is not consistent with acute cardiac dysfunction, patient may not be seen physically. This is in an effort to minimize exposure to COVID+ patients within the healthcare system.
Indications for cardiology consultation in COVID+ Patients:
· New ventricular arrhythmia (Does not include PVCs or NSVT that is not hemodynamically significant)
· New bundle branch block, alternating bundle branch block, or new heart block
· ST elevation on ECG or direct patient history concerning for acute coronary ischemia
· New and acutely decompensated heart failure (left or right ventricular) as evidenced by physical exam and laboratory data.
· Concern for cardiogenic shock.
· Significant worsening in oxygenation or vasopressor requirement
· Request for echocardiogram
Triggers for echocardiography:
Echocardiography is a valuable tool for assessing critically ill patients. However, echocardiograms will invariably increase the risk of inadvertent exposure due to the need for prolonged patient-sonographer interaction and the difficulty with cleaning the echo machines. The risks of exposure must be weighed against the benefit of a study and the expected likelihood of a helpful result. The risks of exposure with echocardiography significantly exceed the risks of exposure with basic bedside physical examination maneuvers and echocardiography should not be used for this purpose. All COVID+ echocardiograms, should be discussed with the echocardiography attending/fellow during weekdays or the consult fellow on weekends/overnight. Echocardiography should only be done if it is expected provide clinical benefit. (link to echo guideline here)
· New ventricular arrhythmia (Does not include PVCs or NSVT that is not hemodynamically significant)
· New bundle branch block, alternating bundle branch block, or new heart block
· New symptomatic heart failure (based on physical exam suggestive of intravascular volume overload)
· New unexplained shock (based on physical exam, laboratory data, and ScVO2 suggestive of cardiogenic cause of shock)
· Patients requiring intubation or ICU admission with rapidly rising and/or significantly elevated troponin and/or BNP
· Request of the cardiology consult service
References:
1. Guan W-J, Ni Z-Y, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. February 2020. doi:10.1056/NEJMoa2002032
2. Li B, Yang J, Zhao F, et al. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin Res Cardiol. March 2020:10.1007/s00392-020-01626-01629. doi:10.1007/s00392-020-01626-9
3. Bonow RO, Fonarow GC, O’Gara PT, Yancy CW. Association of Coronavirus Disease 2019 (COVID-19) With Myocardial Injury and Mortality. JAMA Cardiol. March 2020. doi:10.1001/jamacardio.2020.1105
4. Guo T, Fan Y, Chen M, et al. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. March 2020. doi:10.1001/jamacardio.2020.1017
5. Shi S, Qin M, Shen B, et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. March 2020. doi:10.1001/jamacardio.2020.0950
6. Hu H, Ma F, Wei X, Fang Y. Coronavirus fulminant myocarditis saved with glucocorticoid and human immunoglobulin. Eur Heart J. March 2020. doi:10.1093/eurheartj/ehaa190
7. Inciardi RM, Lupi L, Zaccone G, et al. Cardiac Involvement in a Patient With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. March 2020. doi:10.1001/jamacardio.2020.1096
8. Chapman AR, Bularga A, Mills NL. High-Sensitivity Cardiac Troponin Can Be An Ally in the Fight Against COVID-19. Circulation. April 2020. doi:10.1161/CIRCULATIONAHA.120.047008
9. Thachil J et al. ISTH interim guidance on recognition and management of coagulopathy in COVID19. J Thromb Haemost 2020 Mar 25
ISTH Interim Guidance on Evaluation of Coagulopathy in COVID-19:
Transthoracic and Transesophageal Echocardiograms
COVID-19 UCSF ECHOLAB GUIDELINE
INTRODUCTION:
Based on available evidence and expert opinion, the COVID-19 virus, similar to other coronaviruses and influenza, is primarily transmitted through close contact and infected droplets.
Echocardiographic services will be required in the care of some patients with suspected or confirmed COVID-19. Consequently, echo providers will be exposed to SARS-CoV-2.
The circumstances surrounding the outbreak are, of course, extremely dynamic, and this Guideline is subject to change. We direct echocardiography practitioners to the Centers for Disease Control (CDC) website for the latest updates and recommendations and to follow the UCSF updates.
UCSF HEALTH RECOMMENDATION
Based upon growing global experience, World Health Organization (WHO) guidance, and revised Centers for Disease Control and Prevention (CDC) recommendations, all UC Health medical centers and many other academic medical centers across the U.S. have already or are moving to droplet/contact precautions with eye protection for ambulatory, hospital, and Emergency Department patients with known or suspected COVID-19 disease.
Patients who are rule-out or are confirmed to have COVID-19 requiring aerosol- generating procedures like intubation, high-flow nasal cannula, non-invasive ventilation, and/or tracheostomy will remain in Novel Respiratory Precautions (airborne /contact precautions with eye protection) and in a negative pressure airborne infection isolation room. N95 with eye protection or PAPR should be used in addition to contact precautions. For all other situation regular face mask should be used.
Clinicians should use their judgment to determine if a patient has signs and symptoms compatible with COVID-19.
INDICATIONS FOR ECHOCARDIOGRAPHY
Echocardiograms (TTEs, stress echoes and TEEs) should only be performed if they are expected to provide clinical benefit. Screening of indications may be indicated, particularly for symptomatic patients whose SARS-CoV-2 status is unknown.
Repeat echocardiograms should not be performed unless there has been a clear change in clinical status.
There are echocardiograms that could be safely postponed until a later date. There are two ways to identify these studies:
· Determine which studies are “elective” and reschedule them, performing all others.
· Identify “non-elective” (urgent/emergent) indications and to defer all others.
These two different ways of looking at indications should both prioritize patients who are at risk for significant morbidity or mortality in the short term if echocardiographic exams are not performed.
Workflow for triage of patient:
· Outpatient orders for routine TTE – postpone and if unclear if can be postponed ask ordering provider. If need more help echo fellow or attending can assist.
· Inpatient routine TTE – check for indication and check for COVID status, symptoms and if recent prior study. If questions or concerns ask echo fellow or attending to assist.
· Urgent orders – check for indication and check for COVID status, symptoms and if recent prior study. If questions or concerns ask echo fellow or attending to assist.
ULTRASOUND ENHANCING AGENTS
Plans for ultrasound enhancing agent (contrast) utilization should be made in advance in order to prevent a sonographer having to wait for the agent to be delivered or having to use more personal protective equipment to exit the patient’s room to obtain the agent.
LENGTH OF STUDIES
Prolonged scanning can expose the sonographer to added risk. These studies should not be performed by a sonography student or any other novice/inexperienced practitioner, in order to minimize scanning time while obtaining images of the highest possible quality.
We do recommend full studies rather than focused for most. This will enable us to better understand this novel patient group and aid us in ongoing and future research. Of course safety comes first.
Sonographers should obtain all images as usual but all measurements should be performed off line to avoid increased length of exposure.
SPECIFIC CONSIDERATIONS FOR TEEs
TEEs carry a heightened risk of spread of the SARS-CoV-2 since they can provoke aerosolization of a large amount of virus. TEEs therefore deserve special consideration in determining when and whether they should be performed, and under what precautions (described below).
TEEs should be postponed or canceled if the indication is rarely appropriate, if they are unlikely to change clinical care, and/or if an alternative imaging modality (e.g. off axis TTE views, ultrasound enhancing agent with TTE) can provide the necessary information.
As always the echo fellow is to evaluate the patient and indication.
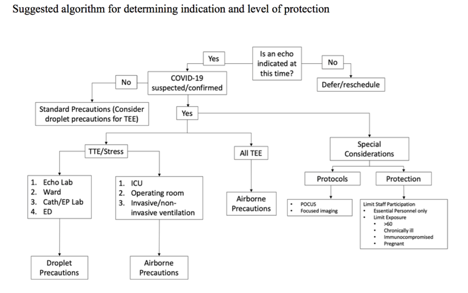
PROTECTION
Figure 1
RISK CATERGORIES (SEE TABLE)
High-risk criteria includes respiratory symptoms and any of the following:
· Travel to identified countries within 14 days
· Contact with someone who has COVID-19
· Requires intubation, other aerosolized generating procedures (e.g., nebulized treatments), or high-flow nasal cannula (high-flow criteria for adult patients)
·
The Safety of the No high-risk approach includes:
· The primary known mode of transmission is via droplet particles
· Exposure to a patient with COVID-19 while wearing droplet (with eye protection) + contact PPE is considered a low-risk exposure by the CDC and internationally.
Appropriate PPE includes:
i. N95 respirator plus face shield/goggles OR powered air purifying respirator (PAPR)
· ii. gloves
· iii. gown
We recommend for now to reuse N95 and Face Shields. Keep the N95 in your pocket and write your name on the Face Shield.
Remember, we need to preserve our higher level PPE (N95, PAPR) for those high-risk patients and procedures! Definitely no N95 for any stress test or transthoracic (unless TEE, COVID +, intubated, or receiving high-flow oxygen).
All TEEs should be performed using N95 and contact precautions unless patient has been ruled out for COVID-19 (assume all may be infected). Airborne precautions should be added for the highest risk patients per UCSF guideline as delineated in the table and figure. Airborne precautions are required during a TEE for suspected and confirmed cases, due to the increased risk for aerosolization. If after evaluation of the patient the attending echocardiologist deems risk higher than indicated by the patient’s care team, the choice can be made to use N95 masks and contact precautions.
If a patient has undergone COVID-19 testing but test results are pending, the procedure should be postponed until results are back.
For stress tests we recommend to use surgical masks and face shields (please re-use face shields by writing your name on the shield).
The ASE recommend shoe covers in addition to the recommendation by UCSF above. We will discuss this with the infection control at UCSF.
The ASE recommends that Staff who are >60 years old, have chronic conditions, are immunocompromised or are pregnant may wish to avoid contact with patients suspected or confirmed to have COVID-19
RISK TABLE:
Criteria | Mask | Eye protection | Contact Precautions | Room type |
High-risk criteria met (Novel Respiratory Isolation) | N95/PAPR | Yes | Yes | Negative-pressure |
No high-risk criteria (Droplet and Contact Isolation) | Surgical | Yes | Yes | Private, standard-pressure, door closed |
No high-risk criteria but COVID-19 testing sent (Novel Respiratory Isolation, standard room) | N95/PAPR | Yes | Yes | Private, standard-pressure, door closed
*Move to high-risk criteria if
|
EQUIPMENT
SARS-CoV-2 is sensitive to most standard viricidal disinfectant solutions.
The Echo machines needs to be cleaned in the room using Clorox Hydrogen Peroxide wipes in any patient on contact precaution including patients who has ruled in for COVID-19 or who are pending COVID-19 results.
Anything left on the machine after the procedure needs to be discarded (electrodes, wipes, etc).
The drawers of the machine should not be opened during the procedure. Items in the drawers, so long as they are closed, are OK.
Wipe down the TEE probe gray bin in the room BEFORE sending it to Endo for processing.
OTHER CONSIDERATIONS
The risk of transmission also occurs in reading rooms.
Keyboards, monitors, mice, chairs, phones, desktops, and door knobs should be frequently cleaned, and ventilation provided wherever possible.
To avoid congregation in the echolab, services wishing to review echos may call the attending on call to review images and go over cases remotely.
RESOURCES
ASE Statement on Protection of Patients and Echocardiography Service Providers During the 2019 Novel Coronavirus Outbreak. ASE 3/18/2020.
UCSF Revised PPE Guidelines, PPE Summary and Rationale for Change. 3/13/2020.
UCSF COVID-19 Control Plan_03-17-2020
Cardiac Catheterization
UCSF Cardiac Catheterization Lab COVID 19 Protocols
Last Updated: March 27, 2020
Introduction:
SARS-CoV2 is the virus that causes COVID-19, a respiratory virus with especially high mortality among the elderly and those with cardiovascular disease. Coronary artery disease in particular appears to increase the risk of death (OR 21.4 95%CI 4.6-98.8).1
Respiratory infections in general confer increased risk for acute myocardial infarction (AMI), with the odds of AMI being increased 6-fold in the week following influenza diagnosis.2 It is likely that SARS-CoV2 will also increase the risk of AMI in vulnerable patients. As such, the cardiac catheterization lab must prepare for a potential influx of patients with COVID-19 infection.
STEMI:
Patients presenting with STEMI during the COVID-19 pandemic should continue to receive primary PCI as the preferred method of revascularization.3 Determination of COVID-19 status will likely be unknown at the time of PCI in STEMI. As such, donning PPE assuming COVID-19 positivity is advised.
Fibrinolytics were used frequently in China for suspected or confirmed COVID-19 cases, but fibrinolytics in general are used more commonly in China than in the United States.4 Fibrinolytics MAY be considered in individual cases of low risk, or stable STEMI (inferior STEMI without RV failure) if limited staffing or equipment resources preclude primary PCI as an initial revascularization strategy.5
NSTEMI:
For most stable NSTEMI patients, there should be sufficient time before angiography to test for COVID-19. Because many patients with COVID-19 can suffer from supply-demand mismatch (Type II MI), it is important to try to differentiate these events from true plaque rupture MI. Deferral of invasive management in select patients is also reasonable if risk is deemed acceptable.
Case selection:
Truly elective cases should be deferred at this time. The definition of “elective” can be challenging and many patients cannot wait indefinitely before needing their cardiac procedure. Those patients in whom the risks of waiting on the procedure are greater than hospital exposure in the midst of the COVID-19 pandemic should be scheduled accordingly.
PPE:
Airborne precautions (N-95 masks) with appropriate eye protection should be used for all suspected and confirmed COVID-19 patients. For all emergent procedures occurring before definitive COVID-19 testing is resulted, similar precautions should be used.
Cath lab location: If there is a major ‘surge’ in the number of COVID cases, consideration should be given for use of one identified lab for COVID positive or PUI and limit the number of staff exposed to these patients.
Intubation: Endotracheal intubation increases the risk of exposure to cath lab staff. In the event that a patient requires intubation in the cath lab, only essential personnel should be present to minimize exposure. If the patient has tenuous respiratory status and is being considered for cath (high O2 requirement, non-invasive positive pressure ventilation), a discussion should be had with ICU/anesthesia teams about consideration of endotracheal intubation prior to call down to the cath lab to reduce the risks of emergent intubation.
1. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. - PubMed - NCBI. https://www.ncbi.nlm.nih.gov/pubmed/?term=Clinical+course+and+risk+factors+for+mortality+of+adult+inpatients+with+COVID-19+in+Wuhan%2C+China%3A+a+retrospective+cohort+study.
2. Acute Myocardial Infarction after Laboratory-Confirmed Influenza Infection. - PubMed - NCBI. https://www.ncbi.nlm.nih.gov/pubmed/29365305.
3. Considerations for Cardiac Catheterization Laboratory Procedures During the COVID-19 Pandemic Perspectives from the Society for Cardiovascular Angi... - PubMed - NCBI. https://www.ncbi.nlm.nih.gov/pubmed/?term=Considerations+for+Cardiac+Catheterization+Laboratory+Procedures+During+the+COVID-19+Pandemic.
4. Fibrinolytic therapy in hospitals without percutaneous coronary intervention capabilities in China from 2001 to 2011: China PEACE-retrospective AMI... - PubMed - NCBI. https://www.ncbi.nlm.nih.gov/pubmed/26787648.
5. Catheterization Laboratory Considerations During the Coronavirus (COVID-19) Pandemic: From ACC’s Interventional Council and SCAI | JACC: Journal of the American College of Cardiology. http://www.onlinejacc.org/content/early/2020/03/16/j.jacc.2020.03.021?_ga=2.64304489.719188611.1585350366-1674237468.1547177882.
ECMO Guidelines for COVID-19
ECMO for COVID-19 Positive Patients: Outside Referrals
The following guidelines are for ECMO indications for COVID patients only. ECMO deployment will be evaluated on a case-by-case basis depending on logistics, resources, personnel, and bed availability. All ICU transfers from outside centers where ECMO is being considered must be discussed with Dr. Aldrich, Dr. Kukreja and other appropriate personal as indicated.
Transfer Protocol:
1) Outside hospital (OSH) requests for transfer of a patient needing advanced respiratory therapy, including ECMO, are forwarded by the Transfer Center to the 6 ICC daytime intensivist or 6/10 ICC nighttime intensivist.
2) The ICC intensivist speaks with the physician managing the patient at the referral center and determines candidacy for advanced respiratory therapy and need for ECMO (see below).
3) The ICC intensivist speaks with Dr. Kukreja and the on-call cannulating surgeon for their approval for candidacy for ECMO.
4) If urgent ECMO is anticipated, the ICC intensivist and on-call cannulating surgeon discuss admission to 6 ICC. Admission to 10ICC will only be considered if a bed cannot be made on 6ICC.
5) On admission to the ICU, the ICU attending and in-house CT surgery provider will evaluate the potential ECMO candidate. The on-call cannulating surgeon and Dr. Kukreja will be contacted by the ICU attending to discuss possible need for and timing of ECMO. If the decision is made to cannulate, the primary team will then become CT surgery.
Candidacy for ECMO in COVID-19 positive patients (see attached ELSO guidelines):
The following five factors will be considered for ECMO candidacy at UCSF:
1) COVID positive;
2) P/F < 100 on FiO2 ≥80% with appropriately high PEEP;
3) Mechanical ventilation < 4 days including at least a trial of prone ventilation, and administration of iNO or epoprostenol if available at referring institution;
4) Age ≤ 60 with minimal co-morbidities and no other failing organ systems;
5) BMI < 35 kg/m2 (if BMI > 30 kg/m2), then referring institution needs to send pictures of the neck and groin to assess potential for cannulation.
Treatment With Renin-Angiotensin System (RAS) Blockers
UCSF Cardiology Division Recommendations for Management Of Patients At Risk for, With Diagnosed, or Suspected COVID-19 Receiving Chronic Treatment With Renin-Angiotensin System (RAS) Blockers
(modified from original document by Mitchell Rosner, Mark Okusa, Li Li, William Petri, James Bergin, Robert Carey, Mohan Nadkarni, Kyle Enfield)
There is concern about risk of increased severity of disease among COVID-19 susceptible and COVID-19 positive patients receiving RAS Blockers. The SARS-CoV2 (COVID-19) virus uses the angiotensin converting enzyme 2 (ACE2) receptor to enter cells. ACE2 is a homolog of angiotensin converting enzyme (ACE). ACE2 negatively regulates the renin angiotensin system by converting Angiotensin II to vasodilatory Angiotensin 1-7, diminishing and opposing the vasoconstrictor effect of angiotensin II. ACE2, ACE, angiotensin II and other renin angiotensin aldosterone system (RAAS) system interactions are quite complex, and at times, paradoxical. Furthermore, tissue expression of ACE2 differ in heart, kidneys and lungs of healthy patients, cardiovascular disease patients and are higher in older individuals. Furthermore, in experimental studies, both ACE inhibitors and ARBs have been shown to reduce severe lung injury in certain viral pneumonias, and it has been speculated that these agents could be beneficial in COVID-19.
While there is an urgent need to study the role of RAS blockers in mediating or moderating Covid-19 susceptibility and disease progression, as of now, given a lack of either large cohort study or randomized trials in this area, we recommend the following:
Recommendations for different clinical groups:
1. Patients with heart failure and reduced ejection fraction
Recommendation: Continue current therapy given the known benefits of RAS blockers in this patient population.
2. Patients with hypertension on RAS Blockers
Recommendation: We endorse the use of RAS blockers for the treatment of hypertension. There is a lack of data on whether patients taking RAS blockers for hypertension should stop these agents and replace them with medications from a different class.
3. Patients with hypertension on RAS Blockers with suspected COVID-19 infection. Recommendation: At this time there is no compelling data to support stopping RAS blockers.
4. Patients with heart failure and reduced ejection fraction and suspected COVID-19 infection.
Recommendation: We recommend continuation of RAS blockers. At this time there is no compelling data to support stopping RAS blockers. We also recommend that these patients should contact their cardiologist and/or primary care physician to discuss their individual case.
5. Patients with confirmed COVID-19 infection
Recommendation: We recommend that consideration be given to stopping RAS blockers in cases of more severe infection and where SIRS and/or hypotension is present. However, this decision should be individualized and made in consultation with the patient’s care team. For critically ill patients with ARDS, acute kidney injury, we recommend discontinuation of RAS blockers.
References
1. Position Statement of the ESC Council on Hypertension on ACE-Inhibitors and Angiotensin Receptor Blockers. March 13, 2020 https://www.escardio.org/Councils/Council-on-Hypertension-(CHT)/News/position-statement-of-the-esc-council-on-hypertension-on-ace-inhibitors-and-ang
2. Hypertension Canada’s Statement on: Hypertension, ACE-Inhibitors and Angiotensin Receptor Blockers and COVID-19
3. Statement of the European Society of Hypertension (ESH) on hypertension, Renin Angiotensin System blockers and COVID-19
https://www.eshonline.org/spotlights/esh-statement-on-covid-19/
4. ACC/HFSA Statement on Ace-I and ARBs
Adult Congenital Heart Disease
UCSF COVID-19 Guidelines for Adults with Congenital Heart Disease
What is COVID-19?
COVID-19 is a newly recognized disease caused by a novel coronavirus that has not previously been seen in humans. The viral infection primarily causes severe respiratory illness, and can be spread from person to person. Information gathered thus far suggests that older adults and those with underlying cardiovascular or pulmonary disease may be susceptible to more severe COVID-19 disease when become infected. This is particularly true for patients with uncontrolled or suboptimally controlled congestive heart failure, hypertension, asthma, COPD, and pulmonary hypertension.
What is known about how COVID-19 affects patients with congenital heart disease (CHD)?
At this early point in the history of the disease, we do not yet have comprehensive data on its behavior in children or adults with congenital heart disease. There is no evidence that having an underlying congenital heart defect increases the chances of being affected. However, based on observations made in older adults with underlying congestive heart failure, it is likely that patients with underlying CHD may be at risk for more severe disease related to the viral infection. It is important to recognize that there is a broad range of anatomical and physiological abnormalities among patients with CHD, and it is likely that not all are at similar risk for severe disease. The UCSF ACHD Program has therefore attempted to classify patients in higher-risk and lower-risk groups. In general, patients with impaired systemic ventricular function and those with abnormal pulmonary blood flow/pulmonary hemodynamics are believed to be at higher risk.
Of note, while asymptomatic hypoxemia is commonly observed in severe COVID-19 disease, ACHD patients with cyanotic heart disease often have low oxygen saturation and low PaO2 at baseline. We recommend physicians review the patient's baseline saturation prior to initiating treatment for asymptomatic hypoxia.
· High risk lesions
· Unpalliated cyanotic congenital heart disease
· Unpalliated shunt lesions with significantly elevated pulmonary blood flow
· Lesions associated with sequestered lung segments (abnormal venous drainage with arterial supply coming from bronchial vessels)
· Fontan physiology
· Cc-TGA
· D-TGA palliated by atrial switch procedures (Mustard or Senning)
· Eisenmenger syndrome or PAH-CHD
· Any lesion with poor systemic ventricular function
· Low risk lesions
· Corrected septal defects and other shunt lesions with no residual defect, absence of pulmonary hypertension, and normal systemic ventricular systolic function
· Palliated tetralogy of Fallot with good ventricular function and competent pulmonic valve
· D-TGA palliated with arterial switch and normal ventricular function
· Isolated BAV or valvar PS with normal ventricular function and no symptoms
At this point, data are insufficient to suggest specific preventive measures in patients with congenital heart disease, and we are recommending that all patients with congenital heart disease, irrespective of which group therein, follow routine public health guidelines with regard to social distancing, scrupulous handwashing, and avoiding unnecessary contact. We have also recommended that whenever possible, patients be allowed to work from home. Patients in the high risk group whose jobs entail work in crowded areas or prolonged close contact with potentially infected individuals (particularly those in healthcare) be allowed to work from home or be excused from work with leave pay.